The life of a human biological sample
March 24th, 2022, by Labtoo's team
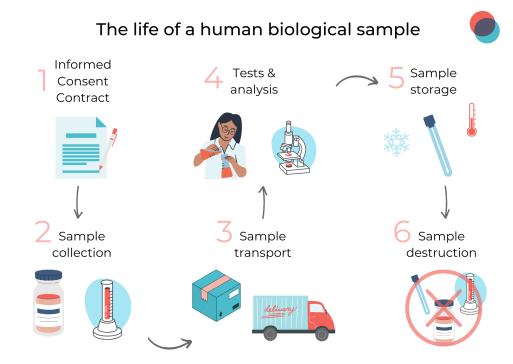
The life of a human biological sample
The first step is to deeply and precisely understand the purpose of collecting a human biological sample before it even exists.
The project must be described in each format that includes the specifications and the expected usage of the biological sample. This is for obvious ethical reasons, since it will remain the property of the human providing the sample, but also because it will determine the regulatory path to take to start collecting.
This will be used all along the life of the samples.
Therefore, the donor will sign the Informed Consent Form or an ICF template will be generated. This englobes the type of sample (blood, tissue, organs…), the frequency, and the uses. The ICF is often mandatory and must be well-determined so that the research can go smoothly.
International review boards, authorities, or ethical committee authorizations are also typically required, depending on the types of samples, the country, and the purpose of the project.
Once all ethical and regulatory authorizations are thoroughly fulfilled, the sample collection at the site can begin. Here, the sample is collected, stored, and packed in the proper way to be shipped to the laboratory. This process is meticulously executed and must answer the previous ICF.
If the samples cross borders, there is a need to engage in an export/import process that could make the transportation last longer. It is wise to prepare this step beforehand so that all can go with no problem.
The fourth step is after the reception of the sample when tests can begin. Here the sample will undergo tests and analysis that will later be compiled in results reports that will be associated with other clinical data, in the Electronic Data Capture system or EDC.
The fifth step can be a rather long one if the samples are kept for future research. The sample is transferred to a biobank where it will be stored for up to 15 to 20 years. If a new study comes up and there is a need for samples, the sample can be retransferred to a new laboratory and used in the new research.
The final step in the life of a sample is destruction. After being used, the sample might be too damaged or old so it will be destructed. The data concerning this sample will be updated to indicate that it is no longer in use and that will close the life cycle for this sample.