Cell culture, ex vivo, and in vitro model services
Cell culture techniques are essential in the development of biotechnology products: because cell culture tests are often the fastest to set up and because there are many different tests: proliferation, migration, viability, inflammation, and apoptosis.
Three main types of cells are used for in vitro testing:
-
immortalized cell lines (a cell that has escaped senescence)
-
primary cell lines (derived directly from an organism, with as few passages as possible)
-
induced pluripotent cells (dedifferentiated stem cells from cultures).
This is why Labtoo developed a tailor-made service to support Labs, Biotechs, Medtech, and Pharma companies in their research program outsourcing.
Finding the most relevant in vitro and ex vivo model for your project
Save time in the experimentation phase
Select your in vitro assays service
Tests under in vitro conditions are carried out in cell culture. These tests allow the study of biological functions and phenomena: each type of test corresponds to a function or mechanism. This applies in particular to cell migration, viability, or immune response. Existing tests are adapted according to projects.
Select your ex vivo assay service
In vitro tests on cultured cells may sometimes have limitations on their representativeness of physiological conditions. In vivo tests on animal models are more complex and expensive to set up and raise ethical questions. Ex vivo tests on tissue explants are a means of testing under conditions closer to the physiological reality.

Select your cell line and primary cells Service
Cell lines are cultured cells from a single clone, derived from tumors, pluripotent cells, or cells taken from an organism and immortalized. These cells serve as models of disease or biological functions, can be reprogrammed into pluripotent cells, or used for bioproduction.
Primary cells are isolated directly from tissues of living organisms and are thus used as models of the physiological state in vivo. Primary culture is not immortal and therefore undergoes the phenomenon of senescence: the aging of the cells. In order to maintain these cells, certain culture conditions must be met.
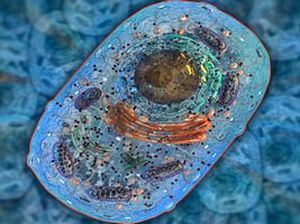
Select your organelles purification service
Organelles are cellular components such as nuclei, mitochondria or ribosomes. The isolation of these compounds is sometimes necessary for their study. For this, several techniques are possible, such as density gradient separation (centrifugation) or affinity purification (chromatography).
Our team will handle your R&D service management from the beginning to the end
Perform a feasibility study by looking for existing expertise available within the network of partners
Set up a study protocol, financial quotation and preparing contracts with lab partners
Implement the study plan into a schedule, collect all needed materials and reagents and execute the service
Do you need more information regarding cell culture and in vitro assay services?
Replacing animal model testing with ex vivo and in vitro models
Each drug development follows a unique process, in terms of drug candidate identification, validation and optimization, pre-clinical and clinical protocols and marketing. There are, however, some broad outlines in the drug development phases, which are described here.
In vivo (from the Latin "in the living") is an experiment using a whole living organism as opposed to a partial or dead organism, or a controlled in vitro environment. Animal experimentation and clinical trials are both forms of in vivo research. In vivo tests are historically used in preference to in vitro tests because they are better suited to observing the overall effects of an experiment on a living subject and may be required in the regulatory framework.
Ex vivo (from the Latin "outside the living") refers to what happens outside an organism. In science, ex vivo refers to experimentation or measurements performed in or on tissues in an artificial environment outside the organism with minimal alteration of natural conditions. A widely used ex vivo study is the chickchorioallantoid membrane test (CAM). Other skin explant studies are commonly used in cosmetic research and development.
In vitro (from the Latin “in glass”) studies in experimental biology are those conducted using components of an organism that have been isolated from their usual biological environment. Cell culture is one type of in vitro models. Three-dimensional (3D) in vitro cell culture has developed greatly in recent years, particularly through the use of gel matrices. 3D culture is more representative of the in vivo environment, especially in cell-cell interactions. Complex in vitro reconstruction models exist, such as reconstructed epidermis models.
Why switching from in vivo to alternative models?
- Developing relevant alternative solutions - a panel of tests to answer today's animal-only questions
- Reduce the costs of complex ex vivo or in vitro models
- Preparing for the transition to the regulatory level
Cell lines: selection criteria
Cell lines are the basis of the research work of many laboratories. Their choice is crucial and must be made in an informed manner, taking into account the following criteria.
The authenticity of the cell line
There are more than 400 misidentified cell lines registered in a specific database. It is then important when you start working with a new cell line, to be sure it is what you think it is. Be sure to obtain your cell line from a trusted cell bank such as ATCC, DSMZ, or ECACC.
Check for contamination
Despite the risk of erroneous or non-reproductive results associated with mycoplasma infection of cell culture, they are often overlooked because of the size of the microorganism and their structural differences from other bacteria. However, many tests exist to verify the absence of mycoplasma: PCR, ELISA, DNA labeling, growth tests, etc.
Prefer a low passage number
Cell lines have a certain genetic instability: at each division, errors become integrated into the genome of the cells, creating differences over time. These differences are regularly revealed by comparing the profiles of equivalent primary and cultured cell lines that have undergone a high number of passages. Good practices include starting from a stock of corresponding low-pass cells on a regular basis (2-3 months, depending on the lineage), and referencing the estimated number of passes in each experiment.
For each biological question, a preferred cell line?
If the biological question can be related to a particular pathology, then it is essential to choose the cell line that will then become the "model" of the disease. But the choice, however critical it may be, is not always obvious. As a starting point for research, the Cancer Cell Line Encyclopaedia (CCLE) provides public access to genetic data for about 1000 cell lines and helps guide choices. The other reference source is the COSMIC (Catalogue of Somantic Mutations in Cancer) database, which also provides data on mutations in cancer cell lines.
If the biological question is independent of a particular pathology, then the reference strains are typically those used in previous research work. However, this approach introduces a bias, since some cell lines are more used than others.
The appropriate scientific solution could be, for example, to test several cell lines on an already validated test, which reinforces the relevance of the observations made on a line used for the whole study.
Find our blog article on HeLa cells to learn more about this cell line with 75 000 scientific articles!
What defines a cell model?
Cell models, if appropriately designed, can be used to rapidly identify the molecular mechanisms of human diseases and develop new therapies.
The design of a robust, reproducible, relatively easy-to-read test (it should use quantifiable molecular markers) is particularly appropriate when using a molecule library for high-throughput screening.
As soon as the model is designed, the experimenter raises the question of how to quantify the expected effect: either by molecular markers or by phenotypic observation.
A molecular marker that is well identified and characterized in a pathology or cellular process has the great advantage of limiting observations linked to an indirect effect.
The FRET technique typically proves to be relevant when studying conformational changes (e.g. intracellular nuclear receptors), aggregation (especially in neurodegeneration), or measurement of receptor-ligand interactions. FRET is based on the transfer of energy between two fluorophores, possible when the two molecules are physically close. Other methods for measuring molecular markers, such as qPCR or dPCR for modulation of gene expression, ELISA or Western Blot for protein quantification, etc., are also available.
Microscopy and imaging in general are useful in phenotypic studies.
Why does a primary culture stop (or continue) growing?
Primary cell culture is the dissociation of cells from an animal or plant parental tissue by enzymatic or mechanical measures and the maintenance of cell growth in a suitable substrate, in glass or plastic containers, under controlled environmental conditions.
Cells derived from primary cultures have a limited lifespan. Cells cannot be kept indefinitely for several reasons. Increasing the number of cells in primary culture will cause depletion of substrate and nutrients. In addition, cell activity will gradually increase the level of toxic metabolites in the culture, which will inhibit cell growth.
The cells can then be resuspended and placed in a new medium, thus eliminating toxic metabolites. This is what constitutes a secondary culture, the goal is to generate a larger number of cells, and to keep the cultures alive. However, there is a risk that the cells may evolve and transform or acquire genetic modifications. The quality of the test will be determined by the capacity for analysis and interpretation.
Used technologies
Cloning and transfection
Organism samples preparation
Microscopy
Plate reader
Separation methods - centrifugation

